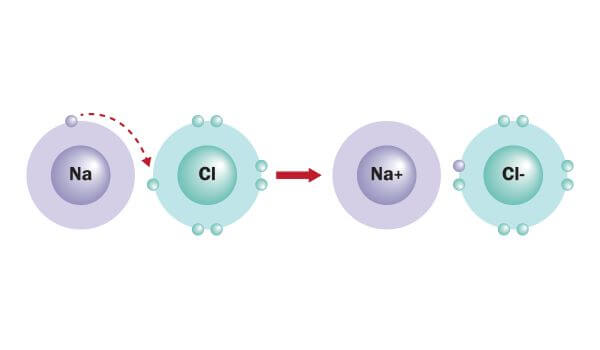
Group 2 they could either gain 6 or lose 2 and as you guessed it there are going to lose those 2 making it a +2 charge. So it's probably going to lose that 1 electron making it a +1 charge. So how do we get that? Group 1 has, could either gain 7 more electrons or lose 1 electron. But if you look at groups 1 through 8 just the representative metals or elements we know that all electrons want to have the same configuration as a noble gas. Okay so how do the other things come together? Well if you look over here we're going, alright these are groups that, these are the representatives of the groups 1 through 8 in the periodic table we're going to exclude the transition metals because those are kind of varying and get a little tricky so we'll talk about those another time. Sodium becomes a positive ion, chlorine becomes a negative ion coming together to what we now know as table salt sodium chloride. Pictorially you can describe this using the Lewis dot diagram sodium has 1 valence electron, chlorine has 7 valence electrons and what happens is sodium is going to give up its electron to chlorine making it up a total of 8 electrons around it and sodium has none which is fantastic. So chlorine has electron configuration of Neon 3S2 3P5 so what happens is sodium is going to say okay "well I'd rather give up this electron so I can have the stable configuration of Neon" and chlorine will like "well I would like to get one so I can have a stable configuration of argon." So what's going to happen? Well the sodium is going to be super nice and give up its electron to chlorine.Ĭhlorine is then going to have a configuration of 3P6 which is great because that's the configuration of argon, very stable and sodium is going to get rid of this and it's going to have a configuration of Neon again very stable so everybody is really happy. Alright so let's take one for example, let's do sodium we know sodium has the electron configuration of Neon 3S1 and that you know it comes with chlorine this is typical table salt the one that you put on your dinner to flavor your food. So they transfer their electrons from the metals to the non-metals.

So what exactly are ionic bonds consisted of? Well they actually make up, metals and non-metals actually make up ionic bonds, metals are cations or which are positively charged ions which means they give off their electrons and actually give their electrons to the non metals which we're going to all the anions or the negatively charged particles. So an ionic bond is a force that holds these ions together in a crystal it's the ratio of ions.
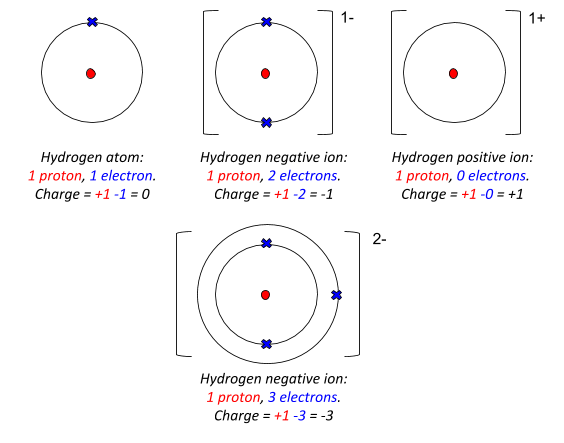
Alright so we're going to talk about ionic bonds, an ionic bond is actually an electric celiac force that holds 2 ions together and don't forget ions are charged particles they actually give or receive extra electrons.

